Why Choose XEOMIN®?
Ingredients Matter—Let’s Talk Tox
By eliminating complexing proteins that can compromise treatment’s effectiveness over time, XEOMIN helps you deliver consistent, repeatable outcomes for upper facial lines.1,2*
Actual patient. Individual results may vary.
*In pivotal trials, no patients demonstrated a secondary lack of treatment response due to neutralizing antibodies.


Real Talk: Is Your Toxin Ready for a Long-Term Relationship?
Fast forward 10 years. Will your toxin require more effort (and dose) to get the same results?
All therapeutic proteins—like those in neurotoxins—have the potential to trigger an immune response. And higher protein loads have been shown to amplify that response by triggering the formation of neutralizing antibodies against the toxin.3,4
Actual patient. Individual results may vary.
Two Ways Neutralizing Antibodies Are Sabotaging Your Relationship
Toxin resistance doesn’t happen overnight. These are the subtle, expensive ways neutralizing antibodies dismantle efficacy—and undermine patient loyalty—over time.
Need for Repeat Injections
36%
of patients who have ever received aesthetic toxin treatment report needing shorter intervals between treatments.5
Increasing Dosage Over Time
66%
of healthcare professionals report needing higher doses or treating more frequently to achieve the same effect.6
Actual patient. Individual results may vary.


In a survey of patients asked about the importance of product purity and consistency5:
57%
of users have experienced diminished effect after 3+ treatments.
74%
of users would switch brands if results started to be less effective.
60%
of users would consider changing providers if decreased effect persists.
Actual patient. Individual results may vary.
A 2023 survey recruited 2,955 toxin users globally to respond to a 25-minute online survey about toxin efficacy. Inclusion criteria were: age 18+ years, history of 3+ treatments, and toxin treatment within the past 12 months. Recruitment was initiated with GenPop sampling to align to census data for age and gender (and in US: region, household income, and ethnicity) to analyze prevalence of toxin users experiencing lack of effect.5
It’s Time to Double Filter Your Standards
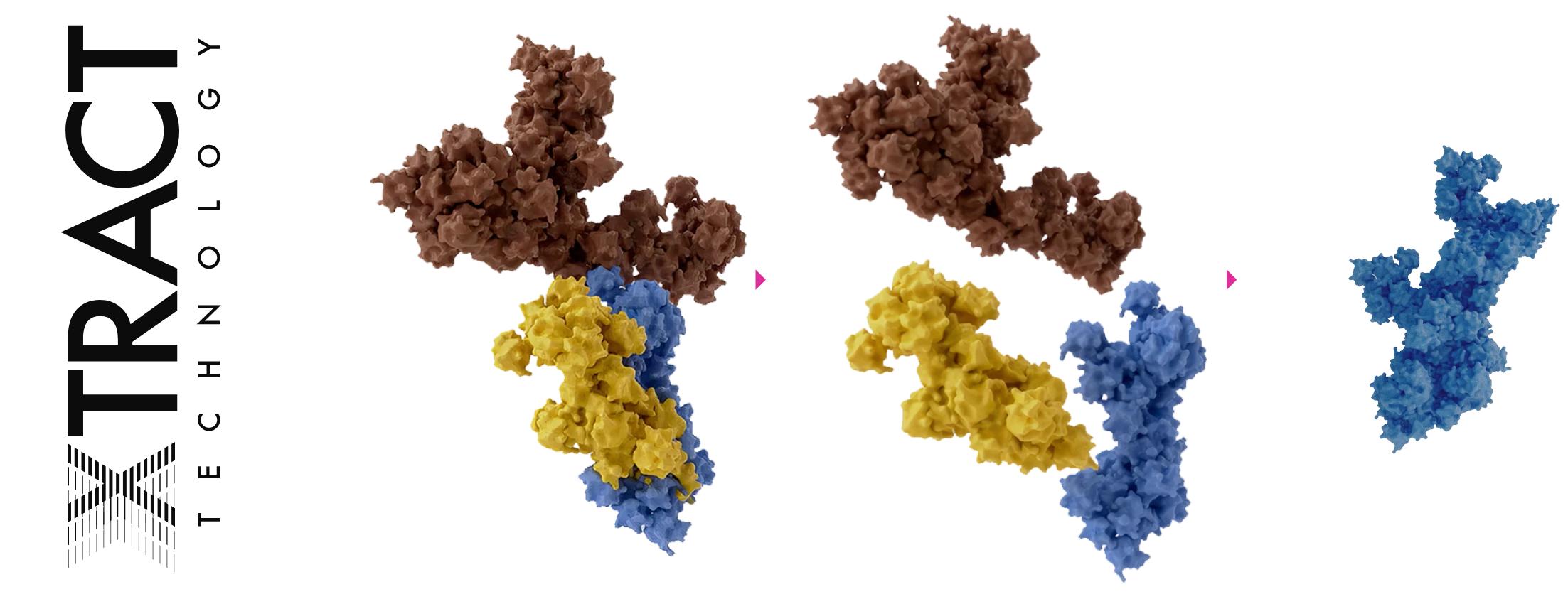
How XEOMIN Works
Only XEOMIN is double filtered with XTRACT TECHNOLOGY®, removing the complexing proteins that can trigger neutralizing antibody formation and reduce efficacy over time.1,2,8,9
Your patients want a long term relationship with their toxin. You want consistent results year after year.
That’s why XEOMIN® is designed for performance dose after dose. It’s the first and only double-purified toxin with XTRACT TECHNOLOGY™ that eliminates complexing proteins and bacterial remnants for consistent results treatment after treatment, and a 96% patient satisfaction rate.
XEOMIN® — Pure. Proven. Performance.
XEOMIN® (incobotulinumtoxinA) is an FDA approved treatment to temporarily improve the look of moderate to severe upper facial lines treated simultaneously or individually.
Effects of XEOMIN may spread hours to weeks after injection, causing serious symptoms. Alert your doctor as difficulty swallowing, speaking, breathing, eye problems, or muscle weakness can be signs of life-threatening conditions.
Side effects may include headache, bruising, injection site reactions, eyelid drooping, brow drooping, and allergic reactions.
Tell your doctor about your medical history, muscle and nerve conditions, and all medicines, especially botulinum toxins, muscle relaxants, and blood thinners, as these may increase the risk serious side effects.
Doses of XEOMIN are not the same as other botulinum toxins.
Reduced Total Protein, Reduced Risk of Antibody Formation
In pivotal trials with over 2,600 patients, XEOMIN delivered results with 0% treatment resistance due to neutralizing antibodies.8
With over 2,000 patients across 30+ real-world studies, evidence continues to show that neutralizing antibodies can impact long-term results.4,10-24
Your choice of toxin matters.
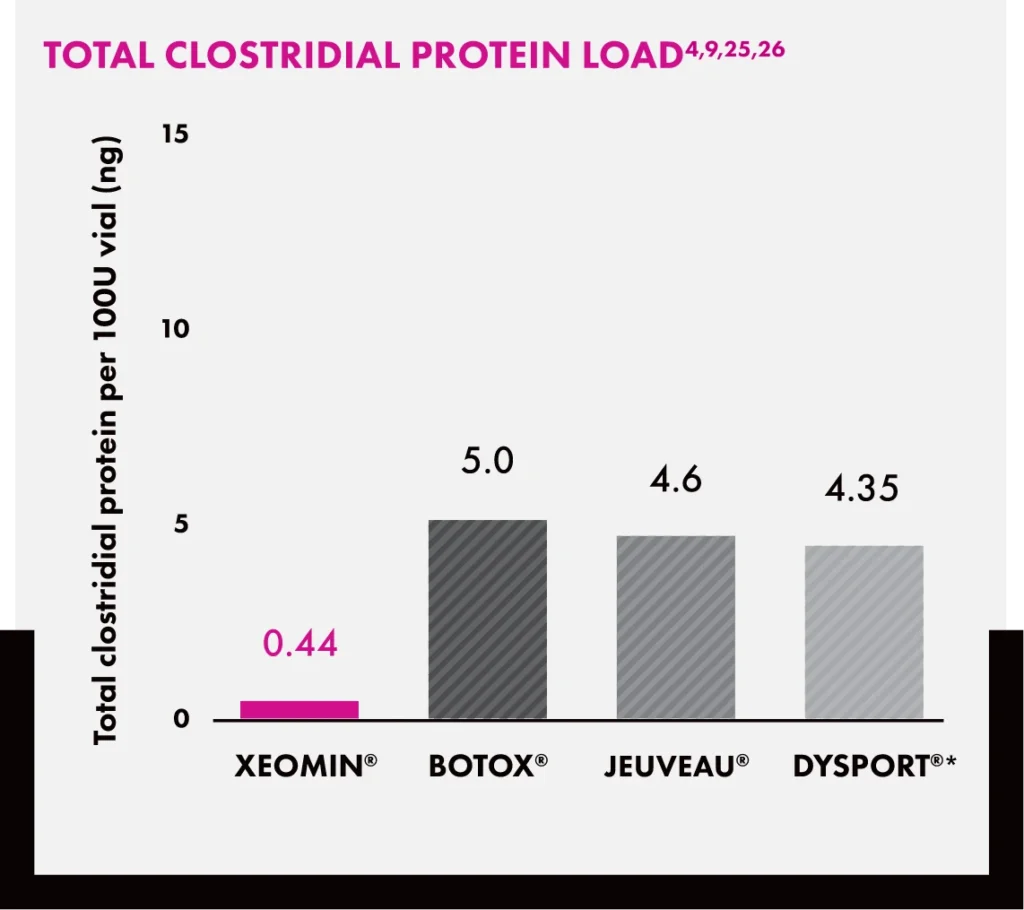
Bar chart titled “Total Clostridial Protein Load” comparing nanograms (ng) of clostridial protein per 100U vial: Xeomin (0.44), Botox® (5.0), Jeuveau® (4.6), and Dysport® (4.35).*
*Total clostridial protein per 100 U (ng) in a 500 U DYSPORT® vial.
These data do not imply differences in clinical performance, safety, or treatment response among products.
Proven to Smooth, Backed by Science
XEOMIN has 15 years of clinical success and global experience.

15 years of real-world aesthetic use27

Approved in 80 countries

20+ million aesthetic treatments globally28
Bring XEOMIN to Your Practice
Your choice of toxin matters. Use XEOMIN to help you stand out and build a practice based on reliable results.

The Proof Behind the Experience
Beautiful results are built on solid science. Explore trial data and peer-reviewed research that supports the XEOMIN experience.