Clinical Evidence for XEOMIN®
Effectiveness Without Excess
In pivotal trials with over 2,600 patients, XEOMIN delivered results with 0% treatment resistance due to neutralizing antibodies.1
Actual patient. Individual results may vary.
Proven Performance for Upper Facial Lines
Treating the full upper facial complex—frown lines, forehead lines, and crow’s feet—can mean using up to 64 units in one session. This higher-unit approach increases patients’ cumulative exposure to neurotoxin, making product purity more important than ever.1
Backed by 15 years of clinical and global experience, XEOMIN is a double-filtered neurotoxin studied in 2 pivotal trials for simultaneous treatment of these 3 key areas of the upper face.2,3
Response Rate at Day 30*: 92.0%3
Dosing: 20 units3
Response Rate at Day 30*: 92.0%3
Dosing: 20 units3
Response Rate at Day 30*: 81.3%3
Dosing: 24 units (12 per side)3
*Based on the investigator-assessed score of none or mild on Merz Aesthetics Scale (MAS) at maximum contraction (secondary endpoint).
Actual patient pictured before treatment with XEOMIN. Individual results may vary.
Results With XEOMIN Lasted Up to 4 Months
Data from 2 upper facial lines trials can help you set patient expectations and plan for return visits to maintain results.
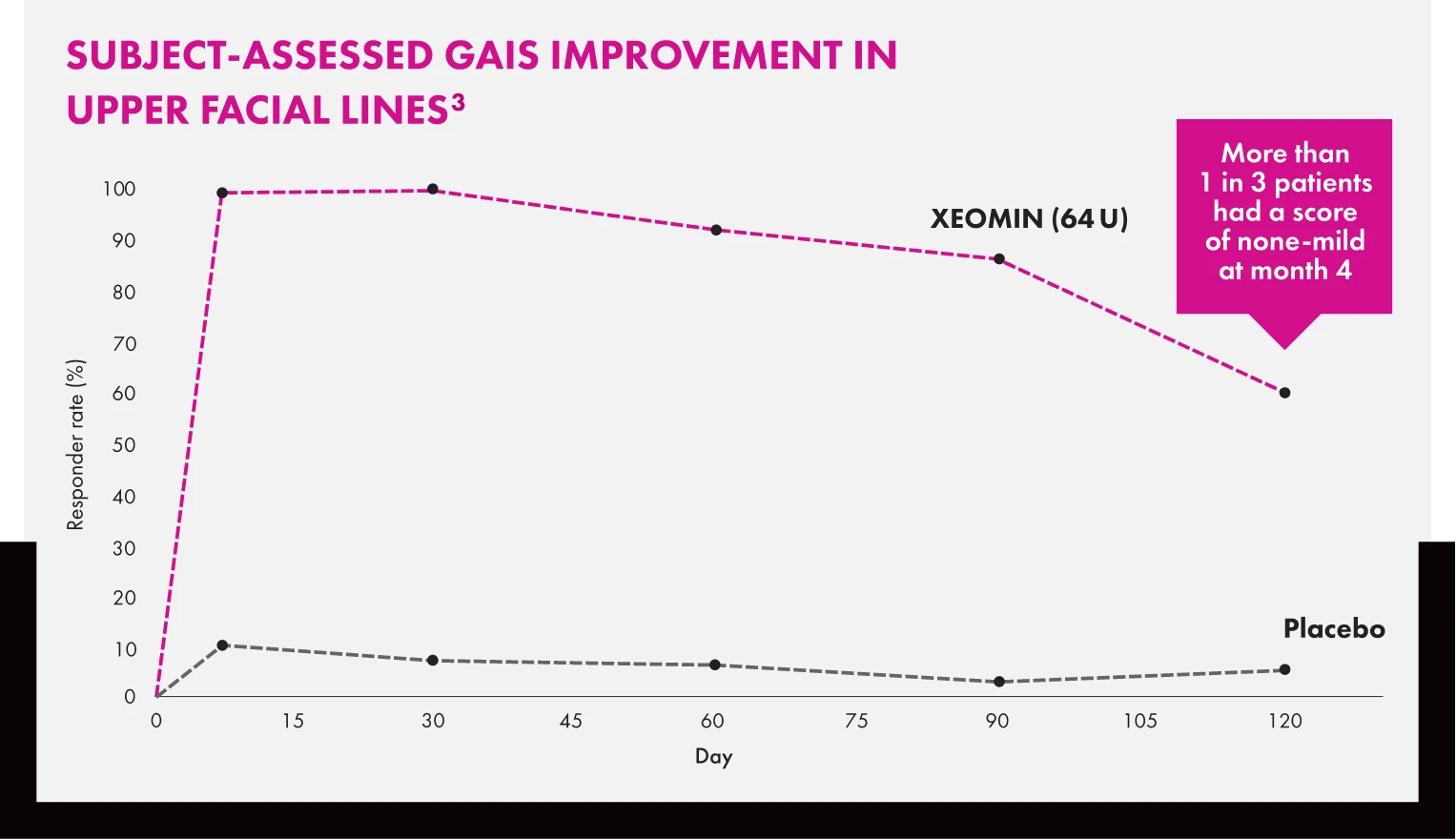
GAIS=Global Aesthetic Improvement Scale.
Line graph titled “Subject-Assessed GAIS Improvement in Upper Facial Lines” showing the responder rate percentage over 120 days. XEOMIN (64 U) peaks at ~100% responder rate on day 30 and maintains approximately ~60% on day 120. The placebo responder rate remains at or below 10% throughout the 120-day period.
XEOMIN’s efficacy and safety for simultaneous treatment of upper facial lines was established in a robust clinical trial program. Over 700 patients were enrolled in two phase 3, randomized, double-blind, multicenter, placebo-controlled studies. Patients were followed for up to a year: Both studies followed patients for up to 120 days and included an open-label extension phase consisting of 2 additional 120-day treatment cycles.
Primary Endpoint: XEOMIN met the primary endpoint of treatment success at Day 30 (composite endpoint of investigator and subject assessment of none or mild and at least a 2-grade improvement on MAS at maximum contraction).

Turn Clinical Evidence Into Clinical Practice
You’ve seen the data—now it’s time to put it to work. Join the thousands of injectors who trust the science behind XEOMIN to deliver consistent, natural-looking results.
Performance in Head-to-Head Clinical Trials
Compared to BOTOX® and DYSPORT® in a study of glabellar lines, XEOMIN® delivered comparable time to onset and duration of effect.*
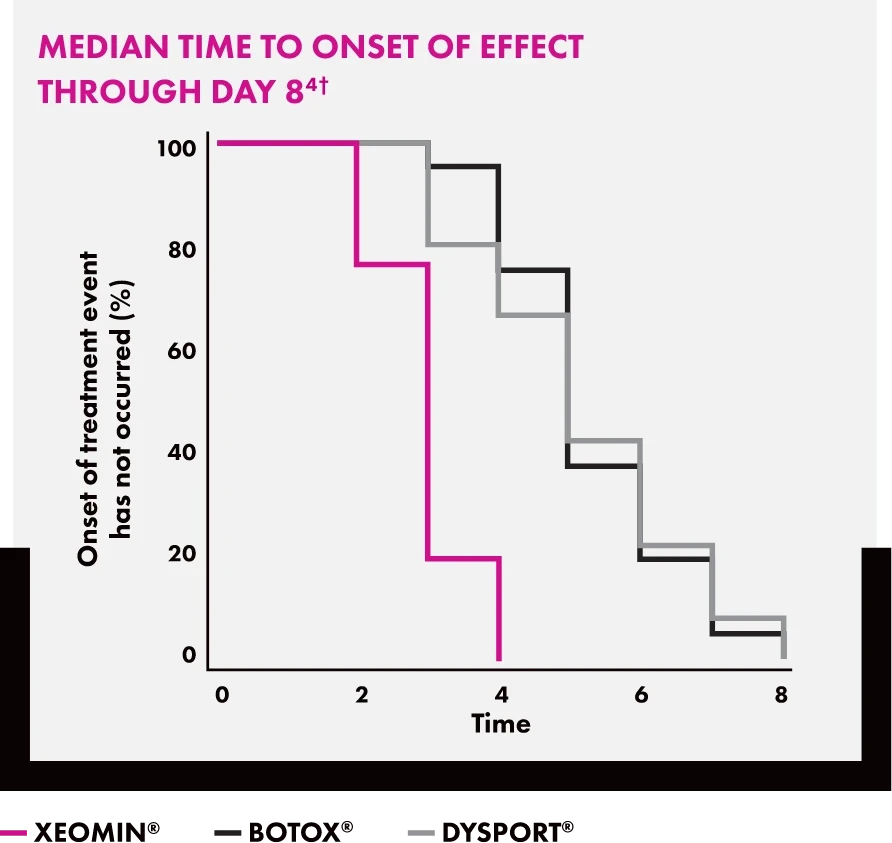
Line graph titled “Median Time to Onset of Effect Through Day 8” measuring the percentage of patients who have not yet experienced onset over an 8-day period. The data show XEOMIN® (magenta) reaching 0% by day 4, while BOTOX® (black) and DYSPORT® (gray) reach 0% by day 8.
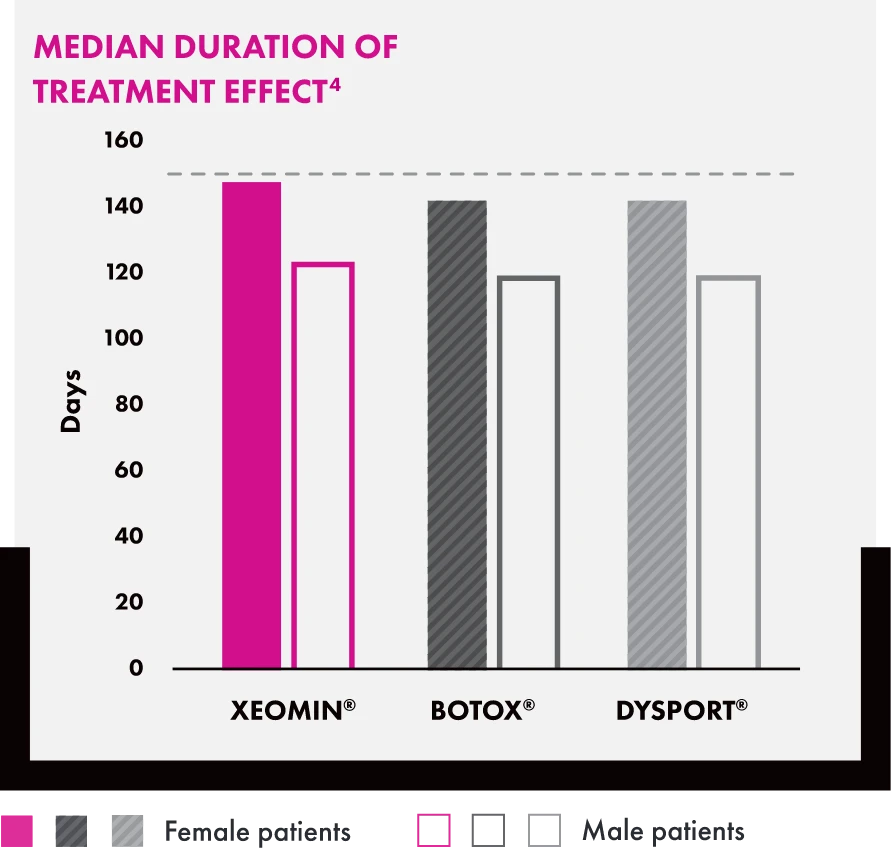
Bar chart titled “Median Duration of Treatment Effect” showing duration in days for female and male patients. Female duration: XEOMIN® (approx. 150), BOTOX® (approx. 140), and DYSPORT® (approx. 140). Male duration: XEOMIN® (approx. 120), BOTOX® (approx. 115), and DYSPORT® (approx. 115).
*Units of biological activity of XEOMIN cannot be compared with or converted into units of any other botulinum toxin products.
†Represents female patients only.
Glabellar Frown Lines Head-to-Head Study Design4
A total of 180 subjects were enrolled (60 per group) in this randomized, controlled study, which directly compared the time to onset and duration of effect for incobotulinumtoxinA (XEOMIN®), onabotulinumtoxinA (BOTOX®), and abobotulinumtoxinA (DYSPORT®) for the treatment of glabellar frown lines.
Subjects received a single treatment of either 21 U incobotulinumtoxinA, 21 U onabotulinumtoxinA, or 63 U abobotulinumtoxinA and were followed for up to 180 days.
Primary Endpoints: Time to onset was defined as the day the investigator noted a decrease in glabellar muscle activity. Duration of treatment effect was defined as the time until muscle action returned to the baseline level.
Head-to-Head Glabellar Frown Lines Trial Design5
In a prospective, multicenter, randomized, double-blind, parallel trial, XEOMIN was randomized 1:1 vs another neurotoxin. The study enrolled 250 healthy female patients (≥18 years old) with glabellar lines of at least moderate severity at maximum frown. XEOMIN demonstrated comparable efficacy and duration up to four months. Patients were classified as responders only if they had a ≥1 point improvement from the baseline on the Facial Wrinkle Scale (FWS) at maximum frown, as assessed by the independent review panel using subject photographs at one month post treatment.
Lasting Effects and Sustained Patient Satisfaction vs BOTOX®
In a head-to-head equivalency study, 250 patients received either 20 units of XEOMIN or 20 units of BOTOX® to treat their glabellar lines.
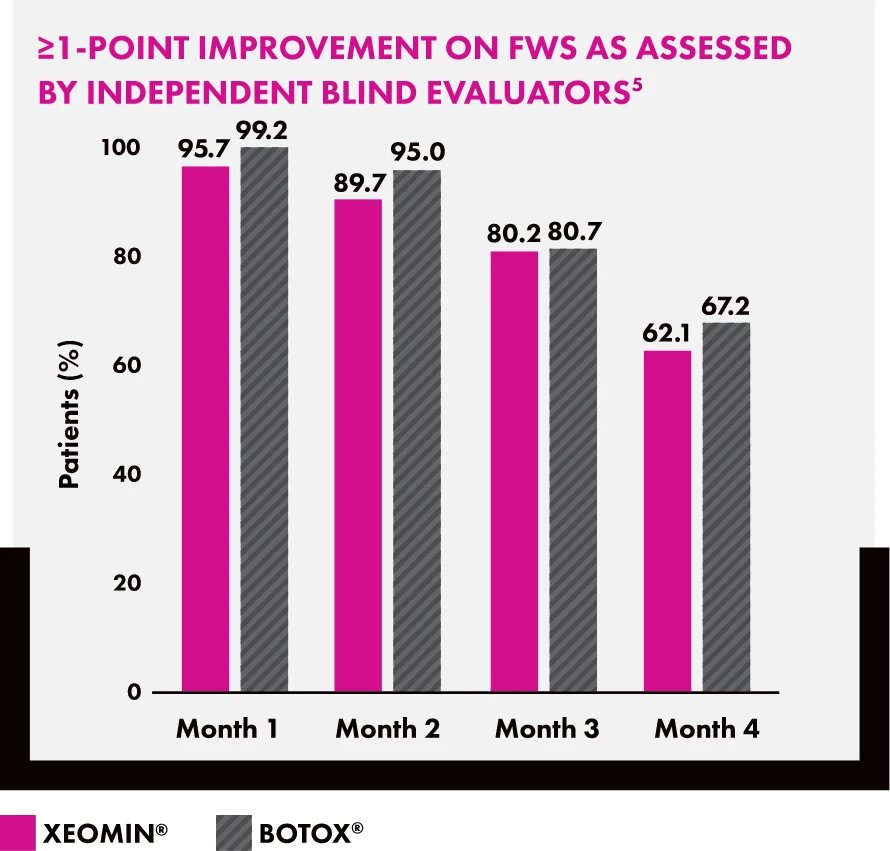
Bar chart titled “≥1-Point Improvement on FWS as Assessed by Independent Blind Evaluators” showing percentage of patients by month. XEOMIN® vs BOTOX®: Month 1 (95.7% vs 99.2%), Month 2 (89.7% vs 95.0%), Month 3 (80.2% vs 80.7%), and Month 4 (62.1% vs 67.2%).
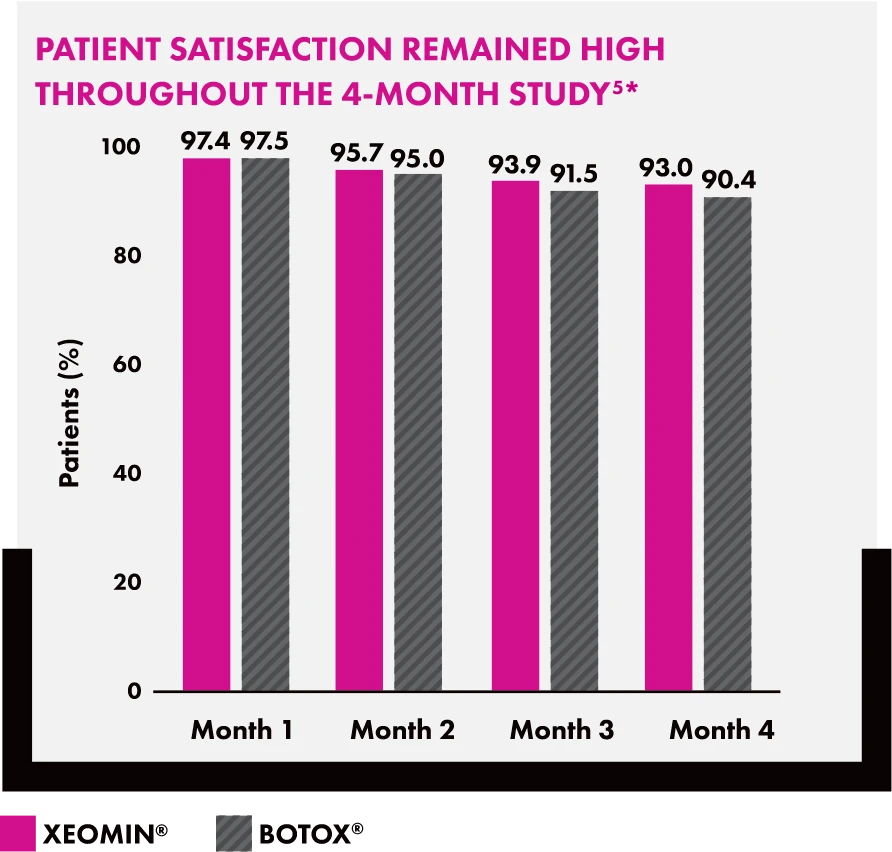
Bar chart titled “Patient Satisfaction Remained High Throughout the 4-Month Study” showing percentage of satisfied XEOMIN® vs BOTOX® patients: Month 1 (97.4% vs 97.5%), Month 2 (95.7% vs 95.0%), Month 3 (93.9% vs 91.5%), and Month 4 (93.0% vs 90.4%).
*Patients responding as “slightly satisfied,” “satisfied,” or “extremely satisfied.”
From Clinical Results to Practical Application
Learn more about reconstituting, dosing, storing, and other practical considerations for using XEOMIN.